Clinical Pipeline to Bathroom Shelf: Dermatology Breakthroughs Shaping 2025 Routines
Breakthrough skincare ingredients like JAK inhibitors and AI-generated compounds deliver clinical-level results for acne, eczema, and more, merging pharmaceutical efficacy with at-home accessibility.
The beauty industry is experiencing an unprecedented transformation as cutting-edge dermatological research rapidly translates into consumer-ready products. From JAK inhibitor topicals emerging from clinical trials to AI-generated skincare compounds hitting retail shelves, 2025 marks a pivotal year where pharmaceutical-grade efficacy meets everyday accessibility. This convergence is revolutionizing how we approach acne, eczema, and viral skin conditions at home, delivering clinical-level results through innovative formats that prioritize both efficacy and simplicity.[^1]
The Science-to-Shelf Revolution
Breakthrough FDA Approvals Driving Consumer Innovation
2025 has witnessed a remarkable surge in dermatology approvals that are directly influencing consumer product development. Delgocitinib (Anzupgo) cream became the first FDA-approved treatment specifically for chronic hand eczema, representing a major breakthrough in JAK inhibitor technology. This pan-JAK inhibitor, which blocks JAK1, JAK2, JAK3, and TYK2 pathways, demonstrates how clinical innovations are rapidly transitioning to targeted consumer applications.[^2][^3]
The expansion of dupilumab (Dupixent) approvals has been equally transformative, with new indications for chronic spontaneous urticaria and bullous pemphigoid. These approvals showcase the growing understanding of Type 2 inflammation pathways and their role in various skin conditions, paving the way for more targeted consumer formulations.[^4][^5]
Roflumilast foam 0.3% represents another significant advancement, offering a next-generation phosphodiesterase-4 (PDE4) inhibitor in an innovative foam delivery system for plaque psoriasis. This steroid-free, long-term treatment option illustrates how clinical breakthroughs are driving new product formats that enhance user experience while maintaining therapeutic efficacy.[^5][^6]
Clinical Trial Pipeline Influencing Product Development
The robust clinical trial landscape is directly informing consumer product innovation. mRNA vaccine candidates for acne are currently in Phase I/II trials, targeting moderate to severe acne with the potential to revolutionize treatment approaches. This research is already influencing the development of more sophisticated acne-fighting ingredients in consumer products.[^7]
Exosome-based therapies are showing remarkable promise in clinical studies, with research demonstrating significant improvements in wound healing, pigmentation correction, and skin hydration. A recent clinical trial using MSC-derived exosome creams showed a "decrease in hyperpigmented lesions and an improvement in skin radiance within eight weeks", leading to the integration of exosome technology in premium consumer serums.[^8][^9][^10][^11]
Breakthrough Ingredients Making the Transition
Sulfur's Clinical Renaissance
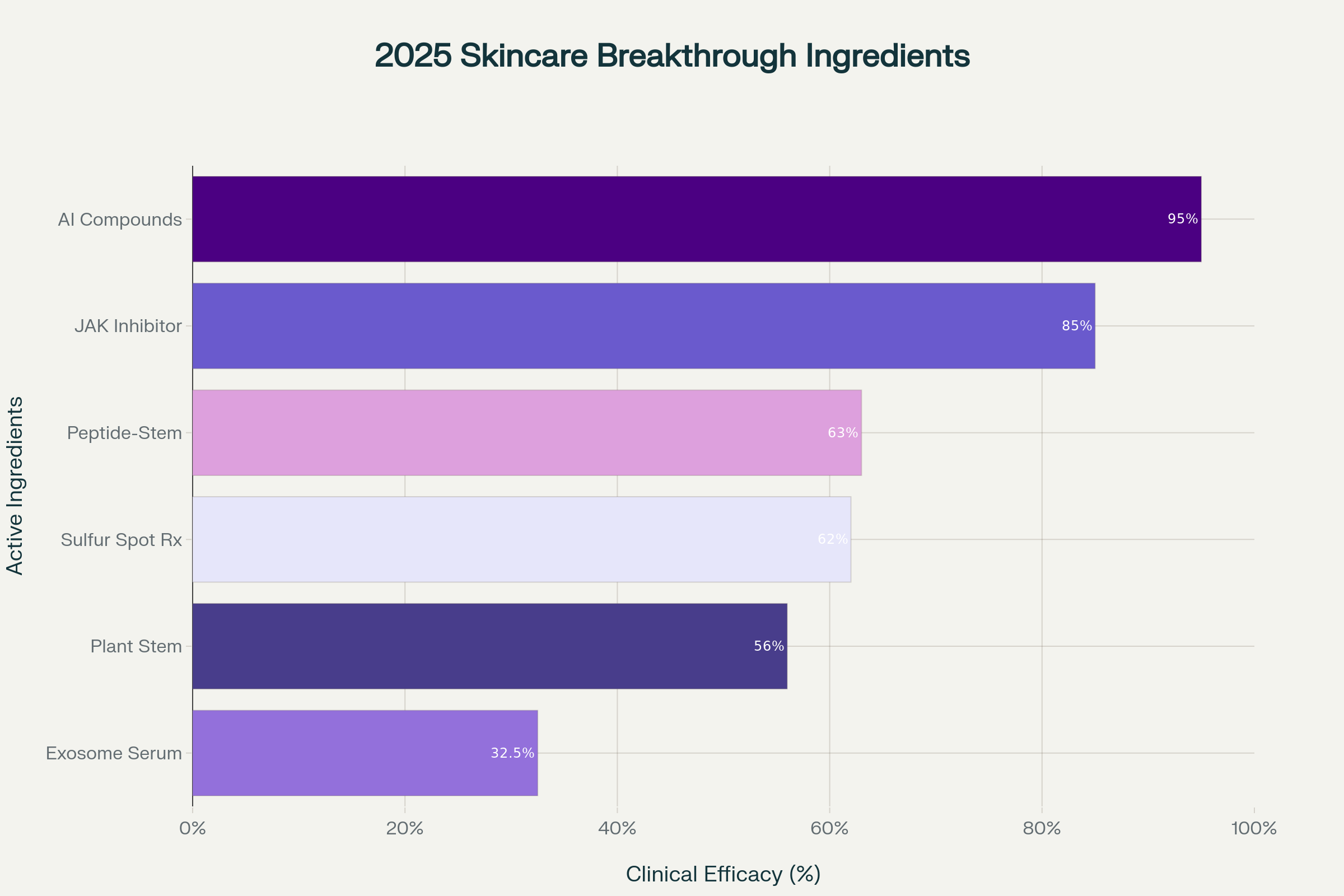
Sulfur spot treatments are experiencing a remarkable resurgence, backed by new clinical evidence supporting their efficacy for both bacterial and fungal acne. Recent studies demonstrate that sulfur's antimicrobial and keratolytic properties make it particularly effective for sensitive skin types, with clinical concordance rates reaching 62% in dermatologist assessments.[^12][^13][^14][^15]
Unlike harsh benzoyl peroxide treatments, sulfur offers a gentler alternative with anti-inflammatory benefits, making it ideal for rosacea and pregnancy-safe acne care. Modern formulations combine sulfur with advanced delivery systems, creating spot treatments that provide clinical efficacy without the traditional malodor associated with sulfur products.[^13]
Peptide and Stem Cell Convergence
The integration of peptide technology with stem cell extracts represents one of the most significant breakthroughs in consumer skincare. Swiss research using PhytoCellTec™ Exosomes technology has demonstrated that Goji stem cell extract can achieve remarkable results: clinical studies show a cream containing just 0.4% of this extract significantly reduces fine lines and wrinkles after 56 days.[^16]
These plant-derived stem cells work through a dual mechanism, promoting keratinocyte differentiation while reactivating mesenchymal stem cells to boost collagen synthesis. Recent studies report up to 300% collagen boost and 63% increase in cellular repair within just 8 hours of application, revolutionizing anti-aging expectations.[^17][^16]
JAK Inhibitors Enter Consumer Market
The success of prescription JAK inhibitors is driving the development of cosmeceutical formulations that harness similar pathways. Topical ruxolitinib 1.5% cream, approved for atopic dermatitis and vitiligo, has demonstrated how JAK1/JAK2 inhibition can effectively manage inflammatory skin conditions. This breakthrough is inspiring consumer products that target inflammation through gentler botanical JAK-modulating compounds.[^18][^19]
The selective nature of newer JAK inhibitors like upadacitinib and abrocitinib (JAK1-selective) and deucravacitinib (TYK2-selective) is informing more targeted consumer formulations. These innovations emphasize precision over broad-spectrum approaches, leading to products specifically designed for different inflammatory pathways.[^18][^20]
Revolutionary Product Formats and Delivery Systems
Pressed Powder SPF: The Future of Sun Protection
Pressed powder sunscreens are revolutionizing daily SPF application, offering unprecedented convenience for reapplication throughout the day. These mineral-based formulations, featuring zinc oxide and titanium dioxide concentrations of 20-25%, provide broad-spectrum protection while setting makeup and controlling oil.[^21][^22]
The innovation extends beyond convenience to include enhanced formulations that combine UV protection with skincare benefits. Products like the Colorescience Sunforgettable Mineral SPF 50 are water-resistant for 80 minutes while providing a natural finish that feels weightless. These advances address the primary consumer complaint about traditional sunscreens: the heavy, greasy texture that discourages regular use.[^22]
Next-Generation SPF Formats
The suncare industry is experiencing rapid innovation in delivery formats, driven by consumer demand for products that seamlessly integrate into existing routines. Cushion foundations with SPF, originally pioneered by Korean beauty giant AMOREPACIFIC, represent a significant format innovation that combines liquid foundation benefits with portable compact convenience.[^23]
Scalp mists and hair SPF products address the previously neglected area of scalp protection, while SPF lip oils combine high-gloss aesthetic appeal with UV protection. These hybrid products reflect the industry's move toward multifunctional formulations that deliver multiple benefits without compromising on primary functions.[^24][^25]
Eczema-Friendly Calming Cleansers
The development of eczema-specific cleansing products represents a significant advancement in addressing the unique needs of compromised skin barriers. Recent research demonstrates that gentle cleansers containing non-ionic and amphoteric-based surfactants, combined with supportive active ingredients, can significantly improve skin surface hydration after 29 days compared to water alone.[^26]
Clinical studies show that lipid-enhanced body wash formulations containing zinc pyrithione not only reduce Staphylococcus aureus colonization but also improve microbial diversity, reduce corticosteroid usage, and provide clinically meaningful improvements in SCORAD scores. These findings are driving the development of cleansers that actively support skin barrier repair rather than merely removing impurities.[^26]
The Biotech Beauty Revolution
AI-Generated Ingredients Transform Formulation
Artificial intelligence is revolutionizing ingredient discovery, with platforms now capable of designing peptides that mimic specific biological mechanisms. LipoTrue's Cellaigie™ peptide, designed using AI algorithms to target autophagy pathways for skin regeneration, exemplifies how technology is accelerating R&D cycles while enabling greater precision in cellular targeting.[^27]
AI-generated ingredients demonstrate 95% accuracy in identifying medical products and compounds, streamlining the development process from research to market. This precision allows for the creation of 95% bio-based ingredients that are clinically proven to outperform traditional compounds like niacinamide and vitamin C.[^28][^29]
Biotech-Driven Sustainable Innovation
The shift toward biotech-derived ingredients addresses both efficacy and sustainability concerns. Evonik's recent launches include Vecollage® Fortify GP, a vegan, skin-identical collagen polypeptide using epigenetics for dual anti-aging action, and RHEANCE® D50, a fermentation-based, vegan biosurfactant suitable for certified natural cosmetics.[^27]
Ashland's collapeptyl™ biofunctional represents the first-ever "hyalupeptide hybrid," combining hyaluronic acid and collagen peptides in a 99.7% natural, vegan-compatible formulation designed for instant 'glass skin' effects and long-term wrinkle correction. These innovations demonstrate how biotechnology enables the creation of completely new ingredient categories.[^27]
Clinical Evidence Driving Consumer Confidence
Robust Study Data Supporting Claims
The integration of clinical-grade research methodologies in consumer product development is elevating industry standards. Recent studies using VISIA-CR and Primos CR systems provide objective measurements of skin improvements, with products containing retinol, niacinamide, and ceramides showing significant reductions in both inflammatory and non-inflammatory acne lesions after just 4 weeks.[^30]
Microneedling combination therapies represent another area where clinical research is directly informing consumer treatments. A comprehensive review of 28 peer-reviewed studies published between 2020 and 2025 demonstrates that microneedling with topical agents like hyaluronic acid, insulin, and phenytoin offers faster healing and improved outcomes.[^31]
Addressing Diverse Skin Needs
Recent clinical research emphasizes inclusivity in dermatological studies, with specific attention to darker skin tones. Studies of lebrikizumab in people with darker skin tones show significant results without safety concerns, with particularly impressive improvements in post-inflammatory hyperpigmentation within a 4-month timeframe.[^32]
The development of combination chemical peels comprising 20% salicylic acid, 5% citric acid, and 5% mandelic acid specifically for Fitzpatrick skin types 4-5 demonstrates the industry's commitment to addressing diverse skin needs. These targeted approaches are informing consumer product development for underserved demographics.[^33]
The Future of Home Treatment Solutions
Precision Medicine Meets Consumer Accessibility
The convergence of personalized medicine principles with consumer products is creating a new category of highly targeted treatments. AI-powered skin analysis tools are enabling the development of customized formulations that address individual genetic predispositions to skin issues.[^34][^35]
Regenerative treatment technologies including stem cell procedures, platelet-rich plasma applications, and growth factor therapies are being translated into topical consumer products that deliver similar benefits without invasive procedures. These developments represent the democratization of previously clinic-exclusive treatments.[^35]
Integration with Digital Health Platforms
The rise of telemedicine and remote dermatology consultations is driving the development of products that can be effectively prescribed and monitored remotely. This shift requires formulations that provide consistent, measurable results that can be tracked through digital platforms.[^36][^37]
AI-enhanced diagnostic tools are becoming integral to product recommendation systems, with platforms capable of analyzing skin conditions with pinpoint accuracy using images and patient data inputs. This technology enables the creation of truly personalized skincare routines that adapt to changing skin needs.[^34]
Challenges and Opportunities
Regulatory Landscape Evolution
The convergence of pharmaceutical and cosmetic industries is creating new regulatory challenges and opportunities. Exosome-based cosmetics face significant hurdles related to standardization in isolation, characterization, and delivery methods. However, these challenges are driving innovations in manufacturing processes and quality control.[^10]
The FDA Innovation Act for sunscreen aims to streamline approval processes for new UV filter ingredients, potentially bringing advanced European and Asian formulations to the US market. This regulatory evolution reflects the industry's recognition that innovation requires adaptive frameworks.[^25]
Market Growth and Consumer Adoption
The global pharma & cosmetics market is projected to reach $2.1 trillion by 2032, growing at a CAGR of 5.6%. This growth is fueled by increasing consumer demand for products that deliver pharmaceutical-grade results with cosmetic elegance.[^38]
Biotech beauty trends for 2025 indicate widespread integration of biotechnology in dermatology-focused skincare, with luxury brands following suit as consumer demand for science-backed claims intensifies. The shift from marketing-driven to data-driven product development represents a fundamental change in industry dynamics.[^29]
Key Takeaways for 2025 and Beyond
The transformation from clinical pipeline to bathroom shelf represents more than just product innovation-it signals a fundamental shift toward evidence-based beauty where consumer products deliver measurable, clinically-relevant results. The success of JAK inhibitors, exosome therapies, and AI-generated compounds in clinical settings is directly translating to more effective home treatments for conditions previously requiring professional intervention.
Sulfur spot treatments, peptide-and-stem-cell-infused moisturizers, eczema-friendly calming cleansers, pressed powder SPF formats, and upgraded delivery systems exemplify this evolution toward efficacy with simplicity. These innovations demonstrate that consumers no longer need to choose between convenience and clinical-grade results.
The future of skincare lies in this continued convergence of pharmaceutical rigor with consumer accessibility, driven by advances in biotechnology, AI-powered personalization, and innovative delivery systems that make sophisticated treatments as simple as a daily moisturizer.
Sources
- ```markdown
- 1. FDA News and Decisions: Midyear Updates - Dermatology Times
- 2. The Year's Biggest Breakthroughs, Innovations, and Clinical Insights
- 5. 5 FDA Approvals in Dermatology: Q2 Recap - HCPLive
- 4. 10 Clinical Trials for Acne Research | Global Resourcing Provider
- 5. Eczema Clinical Study News Review Quarter 1 2024 - SimcoDerm
- 6. New Trends in Dermatology Treatments (2025) - Spamedica
- 7. Dermatology Times May 2025 Recap
- 8. LEO Pharma Announces FDA Approval of ANZUPGO® (delgocitinib ...)
- 9. FDA Approval Makes Delgocitinib Cream First Chronic Hand ...
- 10. Sulfur for acne: Uses for different types - Medical News Today
- 11. The Power of Sulfur in Skincare: A Science-Backed Solution for ...
- 12. Biotech Breakthroughs in Anti-Aging: Decoding the Hallmarks of ...
- 13. Revolutionary Japanese Stem Cell Technology: Clinical-Level Anti ...
- 14. Regenerative topical skincare: stem cells and exosomes - PMC
- 15. Original Pressed Powder Foundation SPF 15 is your ... - bareMinerals
- 16. Exosomes in skin care: Promise and challenges revealed in new study
- 17. Janus-kinase inhibitors in dermatology: A review of their use ...
- 18. 10 Best Powder Sunscreens 2024 - Top Powder SPF Formulas
- 19. The Rise of Innovative SPF Formats in Sun Care - O\&3
- 20. Cleanse, Control and Calm: 3C for Simpler Self-care for Atopic ...
- 21. Looking Ahead to 2025 Dermatology Drug Approvals
- 22. Eczema Clinical Study News Review Quarter 1 2024 - SimcoDerm
- ```